How Top Pharma Companies Evaluate Industry Trends and the Human Element in Containment
ABSTRACT
Engineering controls designed to mitigate exposure and create separation between the operator and the toxic material are available in many different forms, all with their own unique advantages and risks. This article will look into various forms of containment while asking the question, is your liability assured or insured?
ASSURANCE VS INSURANCE
Assurance is defined as a positive declaration intended to give confidence— a promise. We know that nothing is guaranteed in this industry due to the variables involved, but assurance can be provided based on known factors and containment is provided to the level of current risk. On the other hand, insurance is defined as a thing providing protection against a possible eventuality. That is an interesting term, a possible eventuality, as it is an oxymoron. The difference in these terms is the timeline and scope: assurance is limited to known factors in the present, and insurance is both known and unknown moving forward.
In the world of increased potency and operator risk, how can you trust that what will protect you in the present will protect you in the future? Is assurance enough, or is insurance required?
EFFECTIVE CONTROLS?
Besides the industry trend towards increased toxicity, the most threatening risk to containment performance is the operators themselves and their ability to follow GLP (good lab practices) and their given SOP (standard operating procedures). In this case, assurance is simply not enough protection “against a possible eventuality.”
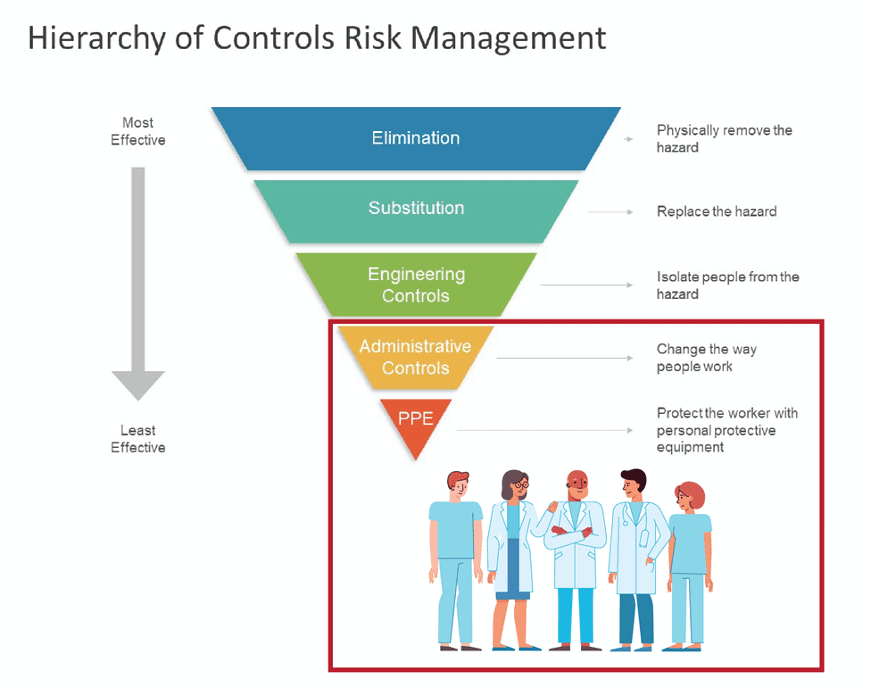
NIOSH’s Hierarchy of Controls prioritizes the most and least effective ways of providing safety. The engineering control is the most effective option when substitution or elimination is not possible. This engineering control is an isolation between the operator and the hazard— in our case, containment systems such as gloveboxes or vented hoods from Flow Sciences, Inc. Decreasing in effectiveness following engineering controls are administrative controls (the way people work) and finally PPE (personnel protective equipment such as coveralls, face masks, etc.…)
What is interesting is that an inanimate barrier is supposedly more effective than the way a capably minded operator performs their work. Think about that for a minute, and really reflect on what NIOSH is saying. The human element is unpredictable, that a barrier is more effective at keeping them safe than their own operating procedures and decisions. The assumed risk of the human element is so evident, it is almost offensive to most sound and intelligent human beings. One would think that the operator would be more capable of making a decision to protect themselves than an inanimate barrier, right? NIOSH says not.
THE HUMAN ELEMENT
In most cases, the human element is celebrated as an advantage to a situation heeding the abilities of humans that machines or equipment cannot provide. In this case, rather, it is viewed with a mostly negative connotation. In lab safety and containment, the human element is a risk that outweighs that of engineering controls and poses a significant dilemma to safety programs.
However, the human element is still important to maintaining an effective containment program. Without operators, the work would not be completed, so training and ease of use for these operators is crucial. As a manufacturer and designer of engineering controls, our responsibility is to provide training and instruction on how to properly use the equipment, but it is ultimately up to the end user to perform their activities to the standards set forth by the safety program.
THE MOST EFFECTIVE SOLUTION
According to NIOSH and the discussions above, the most effective solution is to increase the containment engineering controls to a level beyond the risks of increased toxicity and the human element. The decision between assurance and insurance is as simple as this: should I contain to the minimum of the known factors, or should I anticipate the risks and contain above?
In the chart below, we can see that in typical logic, risk hazard and containment increase linearly for any application where containment is required. This thinking is common, but the top companies in the pharma and biopharma industries are looking at it differently.
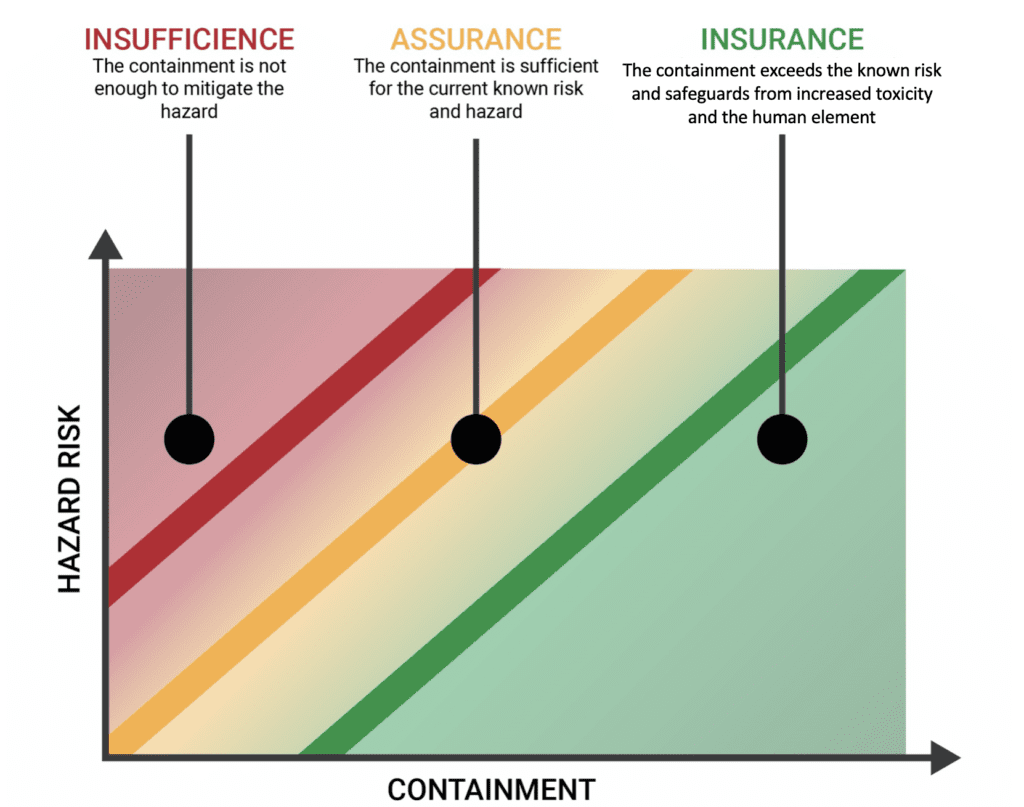
These industries see that the key to growth and scalability is insurance. Not only does this provide protection from potential liability concerns, but it also gives the ability to increase toxicity over time while still protecting their personnel or product.
FINDING CONTAINMENT INSURANCE
Now that we understand the importance, let’s discuss the options. Many systems promote assurance and adequate performance based on the task at hand, but after evaluation these systems do not stand up to the added pressures of the human element, and certainly give no room to increase potency.
In the world today, there are premiere brands that deliver quality product at a price that is above those of lesser quality. We accept this to be true in many cases such as Apple with phones and Porsche with cars. The reason they are of higher quality is because the components and engineering involved costs more than inferior parts and rudimentary engineering. I understand there are outliers, but conventional wisdom tells us that you get what you pay for. While other manufacturers try to minimize manufacturing costs, Flow Sciences has always maintained the goal of maximizing containment performance.
The industry has provided many forms of engineering controls for personnel production that are effective. Respirator programs work well in some situations, but their protection relies heavily on the human element for proper operation and fitting, and PPE programs are also quite expensive programs to maintain. “Soft wall” containment has grown in popularity for its low cost and flexibility, but again, the design is subjective to the human element and can easily be misused or damaged during an application. “Rigid wall” systems are more expensive than soft wall but provide a strong and solid barrier between the operator and the hazard. The industry speaks of the price of rigid wall quite a bit in comparison to soft wall due to the single use aspect, but from an investment standpoint, the cost of a rigid containment system is less over time if, and only if, the system is purchased for insurance, and not assurance.
FLOW SCIENCES, AN INSURANCE PHILOSOPHY
At Flow Sciences, we have many potential customers ask why we are more expensive in some situations than competitors. The answer is simple, and those that understand choose to protect their personnel and product with Flow Sciences containment systems. At Flow Sciences, you get what you pay for, and more. FSI engineers and manufactures containment systems that perform above and beyond the CPT (containment performance target) to provide safety, but moreover, containment insurance.
Many top pharma clients prioritize the effectiveness of their engineering controls by purchasing systems like Hybrid Isolators and Glovebox Workstations that provide a rigid physical barrier between the operator and the hazard. This mitigates and controls the human element even further, as well as giving room to increase potency in the future without having to replace their equipment when that time comes. Being flexible and having that option makes clients adaptable to the market and gives them the highest chance of success in a competitive and time-sensitive industry.

The design and engineering involved in these units have been prioritized to provide containment insurance and flexibility in a marketplace that so badly needs it. Pairing these systems with other containment technologies such as rapid transfer ports and powder transfer valves gives a complete solution for highly potent applications and processes. Click on the above units from Flow Sciences’ newly launched standard product line of Hybrid Isolators and Glovebox Workstations to see specific features and benefits or reach out today to discuss your application and view the hundreds of task specific systems Flow Sciences has engineered. Our containment engineers are available for no-cost consultations to make sure that your engineering control will be the most effective system available, and moreover, provide the containment insurance that you need.
