Fig 1: Perkin Elmer Janus G-3 Liquid Handling System inside a Flow Sciences Custom HEPA Filtered Unit
Unit Mission: Patient Covid-19 Sample Processing
Dr. Robert Haugen
Flow Sciences, Inc.
6/24/20
Recently published scientific papers feature various aspects of pandemic “star” Coronavirus 19. This virus has significantly changed pharma research and production in a variety of unique ways.
This white paper will focus on how research/production efforts might fail under the threats of highly contagious disease viruses, bacteria, and highly active pharmaceutical ingredients. Performing contemporary research safely and producing highly active pharmaceuticals and vaccines will require material isolation and medical product purity. Once identified, contamination paths and bad production procedures will require correction and control.
We will outline possible contamination vulnerabilities in this paper. Flow Sciences and other manufacturers are developing containment options to neutralize such challenges and test methods to certify them. The author will discuss several current enhancements already used and new approaches we are all developing right now!

Where Containment May be Challenged in Today’s Environment:
1.) Collision of Energy Conservation with Containment Challenges
ANSI/AIHA Z 9.5 sets acceptable control levels for chemical fume hoods at stricter levels today than twenty years ago, while stricter energy conservation requirements mandate historically lower exhaust rates for the same equipment in most new facility designs. Recommended containment control levels using ANSI Z 9.5 are now 0.050 ppm as installed1 or lower 2. Twenty years ago, this level was double this value at 0.10 PPM 3.
In spite of achieving mandated containment improvements, a variety of contamination vectors are still possible, particularly with recently reduced exhaust volume requirements established to conserve energy.
2.) Researcher and operator aerosol contamination from Covid 19 carriers:
COVID-19 follows a person-to-person transmission pattern that occurs primarily via direct contact or through droplets spread by talking or coughing from an infected individual. One hundred eighty eight countries have confirmed Coronavirus cases so far 12. Even though transmission vectors are somewhat limited, they work very well for this virus. Not for us humans.
Mass-transportation has also become a vulnerable breeding facilitator13,14. The Diamond Princess Cruise ship harbored in Yokohama, Japan, Holland America’s MS Zaandam cruise ship 6, and various aircraft incidents show how people housed in close quarters can suffer widespread contagion through this limited contact mechanism compounded by a large number of elderly passengers.
Similar to SARS, CoV2, and SARS CoV, the earliest Covid 19 Chinese field studies found active virus in the same rooms as Covid patients 4. In addition, other studies point to contagion in family units 5.
As Covid 19 testing becomes more available, it will be easier to stop infectious people from contaminating lab areas or production facilities by testing, tracing, and isolation. It is also possible to use personal protective equipment (PPE) in particularly sensitive areas.
3.) Measurement of toxic or contaminant leakage outward from a containment device due to poor airflow or electrostatic attraction is less straightforward. For years, the ANSI/ASHRAE 110 test has prescribed diffusion of SF6 gas into chemical fume hood interiors and evaluated quantities that escape containment using a mannequin with an attached air sampler that measured ppm amounts of SF6 in the mannequin-breathing zone.
-
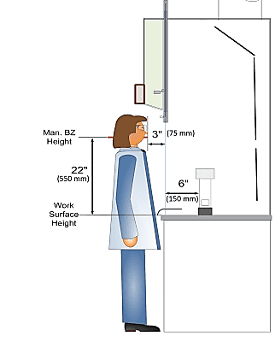
Fig.4 ASHRAE 110 Traditional
-

Fig 5: Standard Test in FSI Test Room
-

Fig 6: ISPE Lactose Containment follows ASHRAE 110
While ASHRAE 110 is not designed to evaluate powder or biological containment, it is nonetheless frequently used for these applications in coordination with a variety of powder tests.

Electrostatically charged powders can stick to the hands of an operator and “hitch a ride” out of the containment area. Air currents may or may not be effective in containing materials exiting the area using this mechanism. Specific additional testing addresses this concern, and FSI has published results comparing ASHRAE 110 tracer gas testing against surrogate powder testing. Results of such tests are beginning to build a comparative record in which we can be confident!

Containment equipment manufacturers generally run ASHRAE 110 tests followed by particulate studies, which provide high-level verification of containment under field conditions. Covid 19 and highly potent pharmaceuticals have only increased the likelihood of using these approaches in tandem to assess containment.
While powder and bio-containment tests developed for pharmaceutical evaluations are today recognized and useful, their conditionality, complexity, and expense have thus far prevented establishment of a broad record of consistent test results from which to build a reference containment standard similar to ASHRAE 110. Unfortunately, it may be next to impossible to generalize powder testing as the industry has done with tracer gas testing as the additional electrostatic vectors of containment escape are so situational that simulated process containment testing (as-used) of particulate escape may be the only answer to confidently apply a given containment approach.
Conclusions:
We do not wish to leave this topic languishing in the swamp of pessimism. The current pandemic has given us several opportunities to use what we have learned containing powders in the related field of viral control.
1.) Good news
- “At the time of publication, no research was available showing that airborne, infective SARS-CoV-2 had been successfully recovered from an air sample in a clinical setting.10”
There appears to be no air-born individual viable virus transmission of Covid 19. This means a developmental containment focus in our field will probably be on very short-range particle transmission. Since manufacturers have good tools available for studying intermediate and room-wide particulate air-born transmission, this industry already has tools for validating product containment in useful and relevant terms using a combination of tracer gas and surrogate powders.
- We present a relevant table of such tools below11:
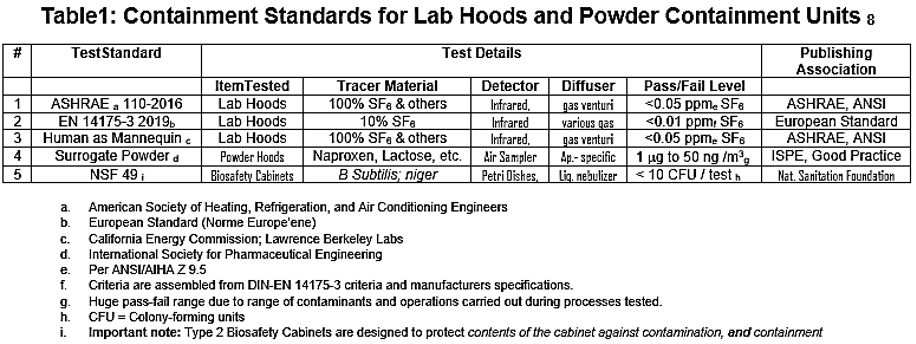
FSI has used most of these tests on a regular basis and they all are relevant today and will probably remain so in the future. While a review of these tests is beyond the scope of this paper, a more thorough discussion is available in the cited reference.
We must never use any single test of containment to validate a research or manufacturing application.
2.) Bad News
While we remain challenged short-term, long-term many elements of our planet lifestyle require remediative changes. Examples include transportation, recreation, entertainment, healthcare, and, of course, safety research itself!
Nevertheless, hiding in this bad news is the key to our own eventual success and improvement. Let us roll up our sleeves and do this work! The future starts yesterday!
Footnotes:
- ANSI/AIHA Z 9.5, Tracer Gas Containment Tests, 2012, P. 72, Section 6.1.2.7
- In most cases because of limitations of SF6 detection, lower containment numbers are attained by running the gas flow rate into the fume hood at 8 lpm rather than 4 lpm. Other steps are used to complicate the hood interior by adding cans and other obstructions (Merck Rahway spec section 115315)
- ANSI/AIHA Z 9.5, Commissioning Plan, 2003, Section 6.2.2
- Aerosol and Surface Distribution of Severe Acute Respiratory Syndrome Coronavirus 2 in Hospital Wards, Wuhan, China, 2020, Zhen-Dong Guo1, Zhong-Yi Wang1, Shou-Feng Zhang1, Xiao Li, Lin Li, Chao Li, Yan Cui, Rui-Bin Fu, Yun-Zhu Dong, Xiang-Yang Chi, Meng-Yao Zhang, Kun Liu, Cheng Cao, Bin Liu, Ke Zhang, Yu-Wei Gao, Bing Lu, and Wei Chen, Abstract
- The characteristics of household transmission of COVID-19, 3/26/2020, Nature Magazine, 17 April 2020, Wei Li, Bo Zhang, Jianhua Lu, Shihua Liu, Zhiqiang Chang, Peng Cao, Xinhua Liu, Peng Zhang, Yan Ling, Kaixiong Tao
- What the cruise-ship outbreaks reveal about CIVUID-19, Nature Magazine, 3/26/2020,
https://www.nature.com/articles/d41586-020-00885-w - Novel COVID-19: A Comprehensive Review of Transmission, Manifestation, and Pathogenesis, 18 May, 2020, Hussain A, Kaler J, Tabrez E, et al.,
https://www.cureus.com/articles/30692-novel-covid-19-a-comprehensive-review-of-transmission-manifestation-and-pathogenesis - Assessing the Particulate Containment Performance of Pharmaceutical Equipment, International Society for Pharmaceutical Engineering, 2012, Chapters 1-8
- Using the ASHRAE 110 Test as a Tool to Improve Laboratory Fume Hood Performance, Hitchings and Maupins, 1997, p.23,
https://www.semanticscholar.org/paper/Using-the-ASHRAE-110-test-as-a-TQM-tool-to-improve-Hitchings-Maupins/4e828cb00c3cc66738f03d9a17f1321637a994d2 - Draft Reference and Guidelines for Post Cleaning Validation of Structures Contaminated with SARS-CoV-2, Indoor Air Quality Association Australia, 6 May, 2020, Bird, Prezant, Green, et.al., section 4.1
- Five Tests for Containment, Flow Sciences, Robert Haugen, 2/27/20, https://flowsciences.com/five-tests-for-containment/
- COVID-19 Dashboard, Johns Hopkins University Coronavirus Resource Center, 23 July 2020, https://coronavirus.jhu.edu/map.html
- Protect Yourself when Using Transportation, Centers for Disease Control, 6/23/2020,
https://www.cdc.gov/coronavirus/2019-ncov/daily-life-coping/using-transportation.html - How to Safely Travel on Mass Transit, Bloomberg City Lab, Simon Clark, Associate Professor of Microbiology at the University of Reading, 6/23/2020,
https://www.bloomberg.com/news/articles/2020-05-27/trains-planes-and-buses-how-to-avoid-coronavirus
